Science Fiction
Dictionary
A B C D E F G H I J K L M N O P Q R S T U V W X Y Z
Healight Ultraviolet Endotracheal Device Has Covid-19 Treatment Potential

The Healight Medical Device Platform Technology is under research development at Cedars-Sinai hospital by Aytu Biomedical.
This medical device technology platform, discovered and developed by scientists at Cedars-Sinai, is being studied as a potential first-in-class treatment for coronavirus and other respiratory infections.Led by Mark Pimentel, MD, the research team of the Medically Associated Science and Technology (MAST) Program at Cedars-Sinai has been developing the patent-pending Healight platform since 2016 and has produced a growing body of scientific evidence demonstrating pre-clinical safety and effectiveness of the technology as an antiviral and antibacterial treatment. The Healight technology employs proprietary methods of administering intermittent ultraviolet (UV) A light via a novel endotracheal medical device. Pre-clinical findings indicate the technology's significant impact on eradicating a wide range of viruses and bacteria, inclusive of coronavirus. The data have been the basis of discussions with the FDA for a near-term path to enable human use for the potential treatment of coronavirus in intubated patients in the intensive care unit (ICU). Beyond the initial pursuit of a coronavirus ICU indication, additional data suggest broader clinical applications for the technology across a range of viral and bacterial pathogens. This includes bacteria implicated in ventilator associated pneumonia (VAP).
"Our team has shown that administering a specific spectrum of UV-A light can eradicate viruses in infected human cells (including coronavirus) and bacteria in the area while preserving healthy cells," stated Dr. Pimentel of Cedars-Sinai. Ali Rezaie, MD, one of the inventors of this technology states, "Our lab at Cedars-Sinai has extensively studied the effects of this unique technology on bacteria and viruses. Based on our findings we believe this therapeutic approach has the potential to significantly impact the high morbidity and mortality of coronavirus-infected patients and patients infected with other respiratory pathogens. We are looking forward to partnering with Aytu BioScience to move this technology forward for the benefit of patients all over the world."
(Via Aytu BioScience Signs Exclusive Global License with Cedars-Sinai for Potential Coronavirus Treatment.)
Science fiction fans recall the idea of healing by some sort of light shown into the body. There are various references to a protoplaser going back to the the original series episode "Space Seed".
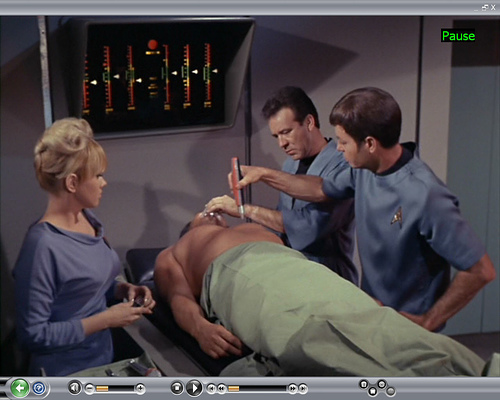
(McCoy uses protoplaser in Space Seed [TOS])
Update 01-Jun-2016: See the article on anabolic protoplaser. End update
She handed him the tiny cutting tool. He made a clean incision through the tissue that had closed in around the barbs, the beam cauterizing as it cut. Then, grasping hold of the short, splintered stub, he gently pulled the head out."Sara, anabolic protoplaser, type zero."
He applied the tip of the instrument to the interior of the wound, slowly working it outward to repair torn veins and gashed arteries, and unite nerves and muscle fibers. Soon, all that was left was the closing of the jagged tear where the spear had gone in and the small incision below it.
"Type two protoplaser."
Scroll down for more stories in the same category. (Story submitted 10/5/2020)
Follow this kind of news @Technovelgy.| Email | RSS | Blog It | Stumble | del.icio.us | Digg | Reddit |
Would
you like to contribute a story tip?
It's easy:
Get the URL of the story, and the related sf author, and add
it here.
Comment/Join discussion ( 0 )
Related News Stories - (" Medical ")
MIT Computerized Bionic Leg Is Part Of The User
'The leg was to function, in a way, as a servo-mechanism operated by Larrys brain, through the mediation of the electronic brain in the leg.' - Charles Recour, 1949.
Bone-Building Drug Evenity Approved
'Compounds devised by the biochemists for the rapid building of bone...' - Edmond Hamilton, 1932.
BrainBridge Concept Transplant Of Human Head Proposed
'Briquets head seemed to think that to find and attach a new body to her head was as easy as to fit and sew a new dress.' - Alexander Belaev (1925)
Natural Gait With Prosthetic Connected To Nervous System
'The leg was to function, in a way, as a servo-mechanism operated by Larrys brain...' - Charles Recour, 1949.
Technovelgy (that's tech-novel-gee!) is devoted to the creative science inventions and ideas of sf authors. Look for the Invention Category that interests you, the Glossary, the Invention Timeline, or see what's New.
Science Fiction
Timeline
1600-1899
1900-1939
1940's 1950's
1960's 1970's
1980's 1990's
2000's 2010's
Current News
FTC: Says Ring Employees Illegally Surveilled Customers
'Then she looked up with a smile and moved closer to the camera.'
Switzerland May Cap Population At Ten Million
'The population of Castle Hagedorn was fixed...'
Project Silica Offers 'Long-Term' Digital Storage
'... folios and tapes and playable discs of platinum alloy.'
Can 'Tactical Umbrellas' Shield One From Drones
'... another corner of his mind began to think about the shields.'
Crystalline Structures In Space, You Say?
A massive space borne lifeform from ST:TNG.
Garçon! A Menu For Artemis II, S'il Vous Plaît
'Michel Ardan, as a Frenchman, was declared chief cook, an important function, which raised no rival.'
Amazing Photonic Crystal Light Sail
'That sail will be twenty thousand miles at the wide part.'
Blue Collar AI Goes To Work To Mine Its Own Crypto
Blue collar bot.
Rogue AI Replicated Itself
'Sapiros computer just kept dialing at random, hanging up on humans, until it got a fellow computer of the same type as itself.'
HandelBot Helps Two-Handed Robots Learn Piano
'I request that you feed the correlation between those dots and the levers of the panel into my memory banks.'
Woven Fiber Electronic Skin For Robots
'... all the feel and appearance of human flesh and epidermis.'
When AI Takes Its First Breath
Any suggestions?
Chinese Aircar Light And Airy, Not For Blade Runners
Daytime version.
The Morphing Wheel And The Smartwheel
'If you surf over a bump, the spokes contract to roll over it.'
Transporting Antimatter
'...drawing plans for the magnetic tongs and bed plates and relays.'
Polish Turns Your Nail Into A Stylus
'He wrote on it, using the pointed fingernail of his right forefinger...'
