Science Fiction
Dictionary
A B C D E F G H I J K L M N O P Q R S T U V W X Y Z
Solar-Powered Fuel Cells Easy As Photosynthesis

A new discovery by MIT scientists lead by Daniel Nocera and Matthew Kanan may make oxygen/hydrogen fuel cells a practical reality.
The key to their discovery is a new catalyst that can produce oxygen gas from water, while another produces hydrogen gas. It consists of consists of cobalt metal, phosphate and an electrode, placed in water. When electricity -- whether from a photovoltaic cell, a wind turbine or any other source -- runs through the electrode, the cobalt and phosphate form a thin film on the electrode, and oxygen gas is produced. The process works at room temperature in neutral pH water; it's easy to set up.
The greatest barrier to the use of fuel cells, which combine hydrogen and oxygen to produce energy and have pure water as a 'waste' product, is the production of hydrogen. Typically, this is done in a way that wastes energy; it burns more energy than it produces.
Nocera and Kanan were inspired by photosynthesis, which is Nature's way of taking the Sun's energy and using it to power the growth of plants and then store that energy.
(MIT energy storage system video w/Nocera interview video)
If this process can be implemented as a consumer product for use in homes, it could make everyone's house a "power plant" and minimizes the need for large power production facilities.
During the daytime, the sun's energy is used to power your house and run MIT's energy process to store energy as hydrogen fuel.
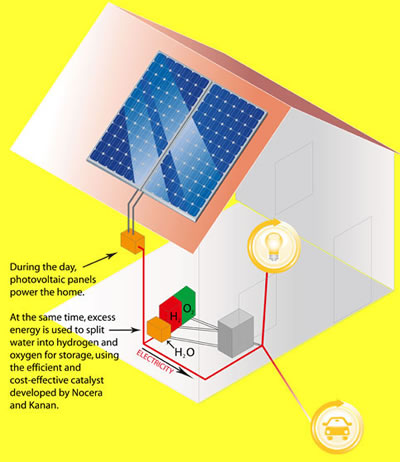
(Store power during the day)
At night, the stored hydrogen can be burned in a fuel cell to provide power for the home, and to charge your electric car for the next day.
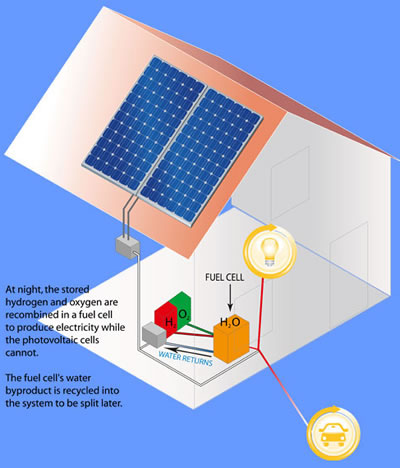
(Use the hydrogen in your fuel cell at night)
Experts are greatly impressed with the technique.
"This is a major discovery with enormous implications for the future prosperity of humankind," said Barber, the Ernst Chain Professor of Biochemistry at Imperial College London. "The importance of their discovery cannot be overstated since it opens up the door for developing new technologies for energy production thus reducing our dependence for fossil fuels and addressing the global climate change problem."
Professor Nocera hopes that this technology will be widely available within ten years for homeowners.
From MIT solar revolution.
Scroll down for more stories in the same category. (Story submitted 8/4/2008)
Follow this kind of news @Technovelgy.| Email | RSS | Blog It | Stumble | del.icio.us | Digg | Reddit |
Would
you like to contribute a story tip?
It's easy:
Get the URL of the story, and the related sf author, and add
it here.
Comment/Join discussion ( 5 )
Related News Stories - (" Engineering ")
Terraformer Industries Make Methane
'Drake was the young spatial engineer he employed to terraform the little rock...' - Jack Williamson, 1942.
Ridiculous 'Ghost Murmur' Tech Still Science Fiction
'...it rears and spreads its fan. It can pick one man out of a crowd.' - Roger Zelazny, 1967.
Infrared Contact Lenses To See In The Dark
'I can see in the dark, Case.' William Gibson, 1984.
Can 'Tactical Umbrellas' Shield One From Drones
'... another corner of his mind began to think about the shields.' - Frank Herbert, 1958.
Technovelgy (that's tech-novel-gee!) is devoted to the creative science inventions and ideas of sf authors. Look for the Invention Category that interests you, the Glossary, the Invention Timeline, or see what's New.
Science Fiction
Timeline
1600-1899
1900-1939
1940's 1950's
1960's 1970's
1980's 1990's
2000's 2010's
Current News
Grok And The City Fathers From 'Cities In Flight' By James Blish
'Chris, the City Fathers are not interested in your welfare; I suppose you know that. They're interested in only one thing: the survival of the city.'
Why Not Move A Warehouse District?
'Did you never see a moving house before?'
Will An AI Found A New Religion?
'You must decide how you will worship Me.'
Terraformer Industries Make Methane
'Drake was the young spatial engineer he employed to terraform the little rock...'
I Need An Outdoor Spherical Display
'Usually a spherical display hovered in the centre...'
Worm Disrupts Physics Simulations Undetected For A Decade
'It diverts integers of the data, the fundamental message-units, so that they no longer agree.'
Muxcard Redditor's DIY Credit Card-Sized Computer
It's a computer, but just barely.
'Soft Assembly' Fashions That Fashion Themselves On The Wearer
'Clothes are no longer made from dead fibers of fixed color and texture that can approximate only crudely to the vagrant human figure...'
Orwell's Nightmare Of AI-Written Novels Comes To Pass
'Books were just a commodity that had to be produced, like jam or bootlaces.'
ISS Plagued By Leak - Again!
'There were perhaps a dozen bladder-like objects in the tunnel...'
Ridiculous 'Ghost Murmur' Tech Still Science Fiction
'...it rears and spreads its fan. It can pick one man out of a crowd.'
Outdoor Video Screens Can Be Arbitrarily Large
The Shape of Things To Come
Infrared Contact Lenses To See In The Dark
'I can see in the dark, Case.'
What'll You Have? Extinct Animals Returned, Or Synthetic Eggshells?
'...a new plastic with the characteristics of an avian eggshell.'
Sunbird Pulsar Fusion Like Leinster's Space Tug
'It was a pushpot, which could not possibly be called a jet plane because it could not possibly fly. Only it did.'
RentAHuman App Lets AI Agents Hire Humans
'She wouldn't stop until Antar had told her everything he knew about whatever it was that she was playing with on her screen.'
